What Would Be the Fate of a Lytic Bacteriophage if the Host Cell Died Prior to the Assembly Stage?
Acellular Pathogens
The Viral Life Wheel
Learning Objectives
- Describe the lytic and lysogenic life cycles
- Describe the replication process of animal viruses
- Describe unique characteristics of retroviruses and latent viruses
- Discuss human viruses and their virus-host jail cell interactions
- Explain the process of transduction
- Depict the replication process of institute viruses
All viruses depend on cells for reproduction and metabolic processes. By themselves, viruses do non encode for all of the enzymes necessary for viral replication. But within a host cell, a virus tin commandeer cellular machinery to produce more than viral particles. Bacteriophages replicate but in the cytoplasm, since prokaryotic cells do non have a nucleus or organelles. In eukaryotic cells, most DNA viruses can replicate inside the nucleus, with an exception observed in the large Deoxyribonucleic acid viruses, such every bit the poxviruses, that tin can replicate in the cytoplasm. RNA viruses that infect animal cells often replicate in the cytoplasm.
The Life Cycle of Viruses with Prokaryote Hosts
The life bike of bacteriophages has been a skillful model for understanding how viruses bear upon the cells they infect, since similar processes take been observed for eukaryotic viruses, which can cause immediate death of the cell or establish a latent or chronic infection. Virulent phages typically pb to the death of the cell through cell lysis. Temperate phages, on the other hand, can become role of a host chromosome and are replicated with the cell genome until such time every bit they are induced to make newly assembled viruses, or progeny viruses.
The Lytic Cycle
During the lytic cycle of virulent phage, the bacteriophage takes over the cell, reproduces new phages, and destroys the cell. T-even phage is a good example of a well-characterized form of virulent phages. There are v stages in the bacteriophage lytic cycle (run into Figure ane). Zipper is the beginning stage in the infection process in which the phage interacts with specific bacterial surface receptors (e.g., lipopolysaccharides and OmpC protein on host surfaces). About phages have a narrow host range and may infect ane species of bacteria or one strain within a species. This unique recognition can be exploited for targeted treatment of bacterial infection by phage therapy or for phage typing to identify unique bacterial subspecies or strains. The second stage of infection is entry or penetration. This occurs through wrinkle of the tail sheath, which acts like a hypodermic needle to inject the viral genome through the cell wall and membrane. The phage caput and remaining components remain outside the bacteria.

The tertiary stage of infection is biosynthesis of new viral components. Afterwards entering the host cell, the virus synthesizes virus-encoded endonucleases to dethrone the bacterial chromosome. Information technology then hijacks the host cell to replicate, transcribe, and translate the necessary viral components (capsomeres, sheath, base plates, tail fibers, and viral enzymes) for the assembly of new viruses. Polymerase genes are usually expressed early on in the cycle, while capsid and tail proteins are expressed afterwards. During the maturation phase, new virions are created. To liberate free phages, the bacterial cell wall is disrupted by phage proteins such every bit holin or lysozyme. The concluding stage is release. Mature viruses flare-up out of the host cell in a process called lysis and the progeny viruses are liberated into the environment to infect new cells.
The Lysogenic Cycle
In a lysogenic cycle, the phage genome as well enters the cell through attachment and penetration. A prime number case of a phage with this type of life cycle is the lambda phage. During the lysogenic cycle, instead of killing the host, the phage genome integrates into the bacterial chromosome and becomes part of the host. The integrated phage genome is chosen a prophage. A bacterial host with a prophage is chosen a lysogen. The process in which a bacterium is infected by a temperate phage is called lysogeny. It is typical of temperate phages to exist latent or inactive inside the cell. As the bacterium replicates its chromosome, information technology as well replicates the phage's DNA and passes information technology on to new daughter cells during reproduction. The presence of the phage may alter the phenotype of the bacterium, since it can bring in actress genes (e.k., toxin genes that can increase bacterial virulence). This change in the host phenotype is called lysogenic conversion or phage conversion. Some bacteria, such as Vibrio cholerae and Clostridium botulinum, are less virulent in the absence of the prophage. The phages infecting these leaner comport the toxin genes in their genome and heighten the virulence of the host when the toxin genes are expressed. In the example of V. cholera, phage encoded toxin can cause severe diarrhea; in C. botulinum, the toxin can cause paralysis. During lysogeny, the prophage will persist in the host chromosome until induction, which results in the excision of the viral genome from the host chromosome. Afterwards consecration has occurred the temperate phage can proceed through a lytic wheel and then undergo lysogeny in a newly infected cell (run across Figure 2).

This video illustrates the stages of the lysogenic life bicycle of a bacteriophage and the transition to a lytic phase.
Think well-nigh It
- Is a latent phage undetectable in a bacterium?
Transduction
Transduction occurs when a bacteriophage transfers bacterial Dna from one bacterium to some other during sequential infections. There are two types of transduction: generalized and specialized transduction. During the lytic cycle of viral replication, the virus hijacks the host cell, degrades the host chromosome, and makes more viral genomes. Equally it assembles and packages Deoxyribonucleic acid into the phage head, packaging occasionally makes a mistake. Instead of packaging viral DNA, it takes a random slice of host DNA and inserts it into the capsid. Once released, this virion volition then inject the old host's DNA into a newly infected host. The asexual transfer of genetic information can allow for DNA recombination to occur, thus providing the new host with new genes (e.thousand., an antibiotic-resistance factor, or a saccharide-metabolizing gene).
Generalized transduction occurs when a random piece of bacterial chromosomal DNA is transferred by the phage during the lytic wheel. Specialized transduction occurs at the finish of the lysogenic bicycle, when the prophage is excised and the bacteriophage enters the lytic cycle. Since the phage is integrated into the host genome, the prophage tin replicate as part of the host. However, some conditions (e.thou., ultraviolet light exposure or chemical exposure) stimulate the prophage to undergo induction, causing the phage to excise from the genome, enter the lytic bicycle, and produce new phages to go out host cells. During the process of excision from the host chromosome, a phage may occasionally remove some bacterial DNA near the site of viral integration. The phage and host DNA from i cease or both ends of the integration site are packaged inside the capsid and are transferred to the new, infected host. Since the DNA transferred past the phage is not randomly packaged but is instead a specific piece of DNA near the site of integration, this mechanism of gene transfer is referred to every bit specialized transduction (see Figure 3). The Deoxyribonucleic acid can then recombine with host chromosome, giving the latter new characteristics. Transduction seems to play an important role in the evolutionary process of bacteria, giving them a machinery for asexual substitution of genetic data.

Recollect well-nigh It
- Which phage life bicycle is associated with which forms of transduction?
Life Cycle of Viruses with Animal Hosts
Lytic beast viruses follow like infection stages to bacteriophages: attachment, penetration, biosynthesis, maturation, and release (see Figure four). However, the mechanisms of penetration, nucleic-acid biosynthesis, and release differ between bacterial and animal viruses. Later on binding to host receptors, creature viruses enter through endocytosis (engulfment by the host jail cell) or through membrane fusion (viral envelope with the host prison cell membrane). Many viruses are host specific, meaning they only infect a certain type of host; and nearly viruses only infect certain types of cells within tissues. This specificity is called a tissue tropism. Examples of this are demonstrated past the poliovirus, which exhibits tropism for the tissues of the brain and spinal string, or the flu virus, which has a primary tropism for the respiratory tract.

Creature viruses do not always limited their genes using the normal flow of genetic information—from Deoxyribonucleic acid to RNA to poly peptide. Some viruses take a dsDNA genome like cellular organisms and tin follow the normal menses. Withal, others may have ssDNA, dsRNA, or ssRNA genomes. The nature of the genome determines how the genome is replicated and expressed as viral proteins. If a genome is ssDNA, host enzymes volition be used to synthesize a second strand that is complementary to the genome strand, thus producing dsDNA. The dsDNA can now be replicated, transcribed, and translated similar to host DNA.
If the viral genome is RNA, a dissimilar mechanism must exist used. There are three types of RNA genome: dsRNA, positive (+) unmarried-strand (+ssRNA) or negative (−) unmarried-strand RNA (−ssRNA). If a virus has a +ssRNA genome, it tin can be translated directly to make viral proteins. Viral genomic +ssRNA acts like cellular mRNA. All the same, if a virus contains a −ssRNA genome, the host ribosomes cannot interpret information technology until the −ssRNA is replicated into +ssRNA past viral RNA-dependent RNA polymerase (RdRP) (see Effigy 5). The RdRP is brought in by the virus and tin can be used to brand +ssRNA from the original −ssRNA genome. The RdRP is also an important enzyme for the replication of dsRNA viruses, because information technology uses the negative strand of the double-stranded genome as a template to create +ssRNA. The newly synthesized +ssRNA copies can and then be translated by cellular ribosomes.
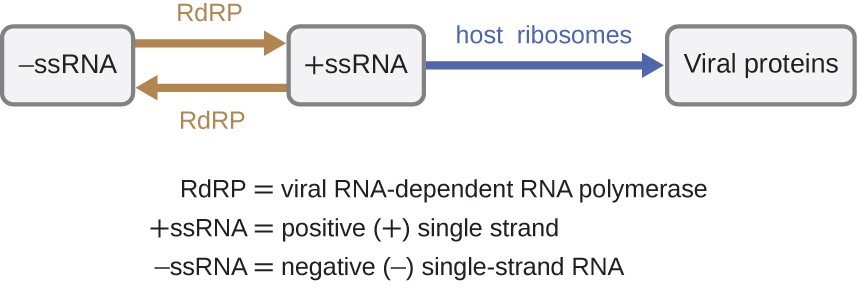
An culling mechanism for viral nucleic acid synthesis is observed in the retrovirus es, which are +ssRNA viruses (see Effigy 6). Single-stranded RNA viruses such as HIV carry a special enzyme chosen reverse transcriptase within the capsid that synthesizes a complementary ssDNA (cDNA) re-create using the +ssRNA genome as a template. The ssDNA is and so made into dsDNA, which tin integrate into the host chromosome and become a permanent role of the host. The integrated viral genome is called a provirus. The virus now can remain in the host for a long time to establish a chronic infection. The provirus stage is similar to the prophage stage in a bacterial infection during the lysogenic wheel. Yet, dissimilar prophage, the provirus does not undergo excision later on splicing into the genome.

Think about It
- Is RNA-dependent RNA polymerase fabricated from a viral gene or a host gene?
Persistent Infections
Persistent infection occurs when a virus is not completely cleared from the system of the host but stays in certain tissues or organs of the infected person. The virus may remain silent or undergo productive infection without seriously harming or killing the host. Mechanisms of persistent infection may involve the regulation of the viral or host gene expressions or the alteration of the host immune response. The 2 chief categories of persistent infections are latent infection and chronic infection. Examples of viruses that crusade latent infections include herpes simplex virus (oral and genital herpes), varicella-zoster virus (chickenpox and shingles), and Epstein-Barr virus (mononucleosis). Hepatitis C virus and HIV are two examples of viruses that cause long-term chronic infections.
Latent Infection
Not all animal viruses undergo replication past the lytic cycle. There are viruses that are capable of remaining hidden or dormant inside the prison cell in a process chosen latency. These types of viruses are known equally latent viruses and may crusade latent infections. Viruses capable of latency may initially crusade an acute infection before condign dormant.
For case, the varicella-zoster virus infects many cells throughout the torso and causes chickenpox, characterized by a rash of blisters covering the peel. Almost 10 to 12 days postinfection, the disease resolves and the virus goes dormant, living within nerve-prison cell ganglia for years. During this time, the virus does not kill the nerve cells or go on replicating. It is not clear why the virus stops replicating within the nervus cells and expresses few viral proteins but, in some cases, typically later many years of dormancy, the virus is reactivated and causes a new affliction called shingles (Effigy 7). Whereas chickenpox affects many areas throughout the torso, shingles is a nerve cell-specific disease emerging from the ganglia in which the virus was fallow.

Latent viruses may remain dormant by existing as circular viral genome molecules exterior of the host chromosome. Others become proviruses by integrating into the host genome. During dormancy, viruses practise non cause any symptoms of affliction and may exist difficult to detect. A patient may exist unaware that he or she is carrying the virus unless a viral diagnostic test has been performed.
Chronic Infection
A chronic infection is a illness with symptoms that are recurrent or persistent over a long time. Some viral infections can exist chronic if the body is unable to eliminate the virus. HIV is an example of a virus that produces a chronic infection, often after a long catamenia of latency. Once a person becomes infected with HIV, the virus can exist detected in tissues continuously thereafter, but untreated patients oft experience no symptoms for years. However, the virus maintains chronic persistence through several mechanisms that interfere with immune role, including preventing expression of viral antigens on the surface of infected cells, altering immune cells themselves, restricting expression of viral genes, and chop-chop irresolute viral antigens through mutation. Somewhen, the damage to the allowed system results in progression of the disease leading to acquired immunodeficiency syndrome (AIDS). The diverse mechanisms that HIV uses to avoid existence cleared by the immune system are also used by other chronically infecting viruses, including the hepatitis C virus.
Think about It
- In what two ways tin a virus manage to maintain a persistent infection?
Life Bike of Viruses with Establish Hosts
Plant viruses are more similar to brute viruses than they are to bacteriophages. Plant viruses may be enveloped or not-enveloped. Like many animal viruses, plant viruses can have either a DNA or RNA genome and exist single stranded or double stranded. All the same, almost plant viruses exercise not have a DNA genome; the majority take a +ssRNA genome, which acts like messenger RNA (mRNA). Just a minority of plant viruses have other types of genomes.
Plant viruses may have a narrow or broad host range. For case, the citrus tristeza virus infects only a few plants of the Citrus genus, whereas the cucumber mosaic virus infects thousands of plants of various plant families. Almost plant viruses are transmitted by contact betwixt plants, or by fungi, nematodes, insects, or other arthropods that act as mechanical vectors. However, some viruses can only be transferred past a specific blazon of insect vector; for example, a detail virus might be transmitted by aphids simply not whiteflies. In some cases, viruses may as well enter salubrious plants through wounds, as might occur due to pruning or weather damage.
Viruses that infect plants are considered biotrophic parasites, which means that they can establish an infection without killing the host, similar to what is observed in the lysogenic life cycles of bacteriophages. Viral infection can exist asymptomatic (latent) or can atomic number 82 to jail cell decease (lytic infection). The life cycle begins with the penetration of the virus into the host prison cell. Side by side, the virus is uncoated within the cytoplasm of the jail cell when the capsid is removed. Depending on the type of nucleic acid, cellular components are used to replicate the viral genome and synthesize viral proteins for assembly of new virions. To establish a systemic infection, the virus must enter a part of the vascular system of the found, such as the phloem. The time required for systemic infection may vary from a few days to a few weeks depending on the virus, the constitute species, and the environmental weather. The virus life cycle is consummate when information technology is transmitted from an infected plant to a good for you institute.
Retrieve about It
- What is the structure and genome of a typical found virus?
Viral Growth Curve
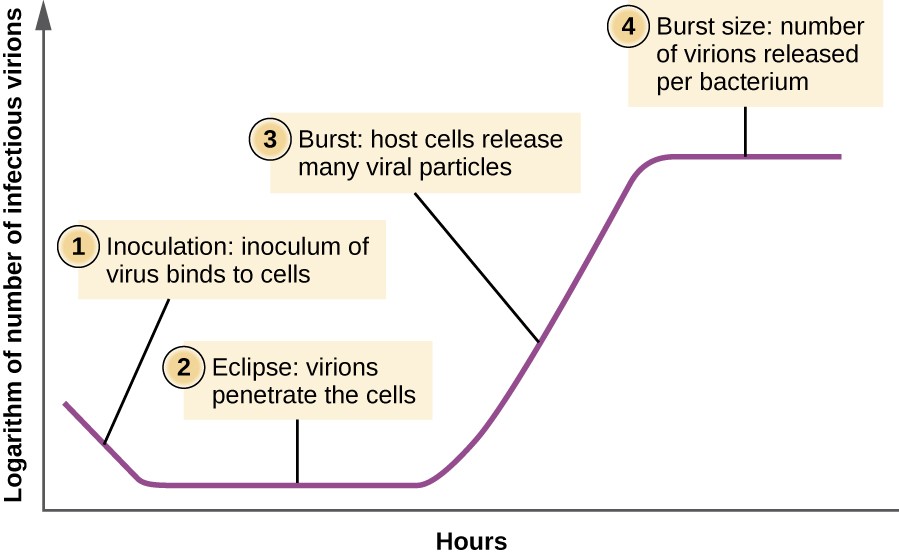
Unlike the growth curve for a bacterial population, the growth curve for a virus population over its life cycle does not follow a sigmoidal curve. During the initial stage, an inoculum of virus causes infection. In the eclipse stage, viruses bind and penetrate the cells with no virions detected in the medium. The chief difference that next appears in the viral growth curve compared to a bacterial growth bend occurs when virions are released from the lysed host cell at the same time. Such an occurrence is called a burst, and the number of virions per bacterium released is described as the burst size. In a one-step multiplication curve for bacteriophage, the host cells lyse, releasing many viral particles to the medium, which leads to a very steep rise in viral titer (the number of virions per unit volume). If no feasible host cells remain, the viral particles brainstorm to degrade during the decline of the civilization (see Figure 8).
Think most Information technology
- What aspect of the life bike of a virus leads to the sudden increase in the growth curve?
Unregistered Treatments
Ebola is incurable and deadly. The outbreak in West Africa in 2014 was unprecedented, dwarfing other human Ebola epidemics in the level of mortality. Of 24,666 suspected or confirmed cases reported, x,179 people died.[1]
No approved treatments or vaccines for Ebola are available. While some drugs have shown potential in laboratory studies and animal models, they have non been tested in humans for safe and effectiveness. Not only are these drugs untested or unregistered but they are also in brusk supply.
Given the dandy suffering and high bloodshed rates, it is fair to inquire whether unregistered and untested medications are better than none at all. Should such drugs be dispensed and, if so, who should receive them, in lite of their extremely express supplies? Is it upstanding to care for untested drugs on patients with Ebola? On the other hand, is information technology ethical to withhold potentially life-saving drugs from dying patients? Or should the drugs perhaps be reserved for health-intendance providers working to contain the disease?
In August 2014, two infected U.s.a. aid workers and a Spanish priest were treated with ZMapp, an unregistered drug that had been tested in monkeys but not in humans. The ii American aid workers recovered, only the priest died. Later that calendar month, the WHO released a report on the ethics of treating patients with the drug. Since Ebola is often fatal, the panel reasoned that it is ethical to give the unregistered drugs and unethical to withhold them for safety concerns. This situation is an example of "compassionate use" outside the well-established organisation of regulation and governance of therapies.
Ebola in the Usa
On September 24, 2014, Thomas Eric Duncan arrived at the Texas Health Presbyterian Infirmary in Dallas complaining of a fever, headache, vomiting, and diarrhea—symptoms commonly observed in patients with the cold or the flu. After examination, an emergency department physician diagnosed him with sinusitis, prescribed some antibiotics, and sent him dwelling house. Two days later on, Duncan returned to the hospital by ambulance. His status had deteriorated and additional blood tests confirmed that he has been infected with the Ebola virus.
Further investigations revealed that Duncan had merely returned from Liberia, one of the countries in the midst of a severe Ebola epidemic. On September 15, nine days before he showed upward at the hospital in Dallas, Duncan had helped transport an Ebola-stricken neighbor to a hospital in Liberia. The hospital continued to treat Duncan, but he died several days after being admitted.

The timeline of the Duncan example is indicative of the life cycle of the Ebola virus. The incubation time for Ebola ranges from 2 days to 21 days. Nine days passed betwixt Duncan's exposure to the virus infection and the advent of his symptoms. This corresponds, in role, to the eclipse menstruum in the growth of the virus population. During the eclipse phase, Duncan would have been unable to transmit the disease to others. However, once an infected individual begins exhibiting symptoms, the affliction becomes very contagious. Ebola virus is transmitted through direct contact with droplets of bodily fluids such every bit saliva, blood, and vomit. Duncan could conceivably have transmitted the disease to others at any time later on he began having symptoms, presumably some time before his arrival at the hospital in Dallas. Once a infirmary realizes a patient like Duncan is infected with Ebola virus, the patient is immediately quarantined, and public health officials initiate a back trace to place anybody with whom a patient like Duncan might have interacted during the menstruation in which he was showing symptoms.
Public health officials were able to track downward 10 loftier-run a risk individuals (family members of Duncan) and fifty low-risk individuals to monitor them for signs of infection. None contracted the illness. All the same, ane of the nurses charged with Duncan'due south care did go infected. This, along with Duncan'southward initial misdiagnosis, fabricated it articulate that U.s. hospitals needed to provide additional training to medical personnel to prevent a possible Ebola outbreak in the US.
- What types of training can ready health professionals to contain emerging epidemics like the Ebola outbreak of 2014?
- What is the departure between a contagious pathogen and an infectious pathogen?
Fundamental Concepts and Summary
- Many viruses target specific hosts or tissues. Some may have more than 1 host.
- Many viruses follow several stages to infect host cells. These stages include attachment, penetration, uncoating, biosynthesis, maturation, and release.
- Bacteriophages have a lytic or lysogenic cycle. The lytic cycle leads to the death of the host, whereas the lysogenic wheel leads to integration of phage into the host genome.
- Bacteriophages inject Dna into the host cell, whereas animal viruses enter by endocytosis or membrane fusion.
- Animal viruses can undergo latency, similar to lysogeny for a bacteriophage.
- The majority of plant viruses are positive-strand ssRNA and tin can undergo latency, chronic, or lytic infection, as observed for animal viruses.
- The growth curve of bacteriophage populations is a 1-step multiplication curve and not a sigmoidal curve, as compared to the bacterial growth curve.
- Bacteriophages transfer genetic information between hosts using either generalized or specialized transduction.
Multiple Choice
Which of the post-obit leads to the destruction of the host cells?
- lysogenic bike
- lytic cycle
- prophage
- temperate phage
[reveal-answer q="294386″]Show Answer[/reveal-answer]
[subconscious-answer a="294386″]Reply b. The lytic bicycle leads to the destruction of the host cells.[/hidden-respond]
A virus obtains its envelope during which of the post-obit phases?
- attachment
- penetration
- assembly
- release
[reveal-answer q="706840″]Show Answer[/reveal-answer]
[hidden-answer a="706840″]Respond d. A virus obtains its envelope during release.[/hidden-reply]
Which of the post-obit components is brought into a jail cell by HIV?
- a Deoxyribonucleic acid-dependent Deoxyribonucleic acid polymerase
- RNA polymerase
- ribosome
- reverse transcriptase
[reveal-answer q="147274″]Show Answer[/reveal-answer]
[hidden-answer a="147274″]Respond d. Reverse transcriptase is brought into a cell by HIV.[/hidden-respond]
A positive-strand RNA virus:
- must kickoff be converted to a mRNA before it can exist translated.
- tin exist used directly to translate viral proteins.
- will be degraded by host enzymes.
- is not recognized by host ribosomes.
[reveal-answer q="761981″]Testify Respond[/reveal-respond]
[subconscious-reply a="761981″]Reply b. A positive-strand RNA virus can be used straight to translate viral proteins.[/hidden-reply]
What is the name for the transfer of genetic information from one bacterium to another bacterium by a phage?
- transduction
- penetration
- excision
- translation
[reveal-respond q="260859″]Show Answer[/reveal-answer]
[hidden-answer a="260859″]Answer a. Transduction is the proper name for the transfer of genetic data from one bacterium to another bacterium by a phage.[/hidden-answer]
Fill in the Bare
An enzyme from HIV that can make a copy of Deoxyribonucleic acid from RNA is chosen _________________.
[reveal-answer q="762404″]Show Answer[/reveal-respond]
[hidden-answer a="762404″]An enzyme from HIV that tin can make a copy of DNA from RNA is called reverse transcriptase.[/hidden-answer]
For lytic viruses, _________________ is a phase during a viral growth curve when the virus is not detected.
[reveal-reply q="889994″]Show Reply[/reveal-answer]
[hidden-answer a="889994″]For lytic viruses, eclipse is a phase during a viral growth curve when the virus is not detected.[/subconscious-reply]
Think virtually Information technology
- Briefly explain the difference between the mechanism of entry of a T-even bacteriophage and an animal virus.
- Discuss the deviation between generalized and specialized transduction.
- Differentiate betwixt lytic and lysogenic cycles.
- Bacteriophages have lytic and lysogenic cycles. Discuss the advantages and disadvantages for the phage.
- How does opposite transcriptase assistance a retrovirus in establishing a chronic infection?
- Discuss some methods by which found viruses are transmitted from a diseased found to a healthy one.
- Label the five stages of a bacteriophage infection in the effigy:

Source: https://opentextbc.ca/microbiologyopenstax/chapter/the-viral-life-cycle/
0 Response to "What Would Be the Fate of a Lytic Bacteriophage if the Host Cell Died Prior to the Assembly Stage?"
Post a Comment